Indicators
Indicators are things that tell you if a substance is an acid, a base or neutral. Below are some of the most common indicators used.
Litmus paper
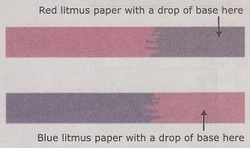
Litmus paper comes in two colors, red and blue, and in useful for finding out if your solution is an acid or a base although it doesn't tell you the strength. To use litmus paper you just dip the bottom of a strip into your solution. Red litmus paper will turn blue in a base but will stay red in a acid or neutral solution. Blue litmus paper will turn red in an acid but stays blue in a base or a neutral solution. So if you test a solution with one color paper and it changes color you don't have to do another test. If it doesn't change color though you have to test it will the other color paper to test if the solution is possibly neutral.
Litmus paper comes in two colors, red and blue, and in useful for finding out if your solution is an acid or a base although it doesn't tell you the strength. To use litmus paper you just dip the bottom of a strip into your solution. Red litmus paper will turn blue in a base but will stay red in a acid or neutral solution. Blue litmus paper will turn red in an acid but stays blue in a base or a neutral solution. So if you test a solution with one color paper and it changes color you don't have to do another test. If it doesn't change color though you have to test it will the other color paper to test if the solution is possibly neutral.

pH Strips
pH strips work similarly to litmus paper, but have the added benefit of being able to determine the pH of your solution. To use a pH strip you dip the end with the three square into the solution so that all three are covered. You then take out the strip and compare it to the key on the box to find the most similar color match. This gives you a number value of the pH.
pH strips work similarly to litmus paper, but have the added benefit of being able to determine the pH of your solution. To use a pH strip you dip the end with the three square into the solution so that all three are covered. You then take out the strip and compare it to the key on the box to find the most similar color match. This gives you a number value of the pH.

Universal indicator
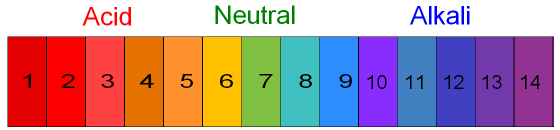
Universal Indicator is a liquid indicator that that, while it won't give you an exact number, can show the the relative strength of your solution. Universal Indicator can be made out of red cabbage. To use Universal indicator you add a few drops to the solution and see what color it turns. Universal indicator will be red in a strong acid, purple in a strong base, and green in a neutral solution. Check out the picture below to see how the color changes with different pH values
Universal Indicator is a liquid indicator that that, while it won't give you an exact number, can show the the relative strength of your solution. Universal Indicator can be made out of red cabbage. To use Universal indicator you add a few drops to the solution and see what color it turns. Universal indicator will be red in a strong acid, purple in a strong base, and green in a neutral solution. Check out the picture below to see how the color changes with different pH values

Phenolphthalein
Phenophthalein is another liquid indicator that is similar to Universal Indicator. This is the indicator that we use in titrations as its color change is very quick and obvious. In a base phenolphthalein turns pink and in a neutral solution it's clear. To use this indicator you just add a few drops to your liquid solution.
Phenophthalein is another liquid indicator that is similar to Universal Indicator. This is the indicator that we use in titrations as its color change is very quick and obvious. In a base phenolphthalein turns pink and in a neutral solution it's clear. To use this indicator you just add a few drops to your liquid solution.
Buffers
Definition of a Buffer
A buffer is a chemical solution that is resistant to changes in its pH as its pH is very stable.
What makes up a Buffer and how does it work?
A buffer is made by adding large volumes of a weak acid or base to its conjugate acid or base as they can be in the same solution with out neutralizing each other. This ensures that if any acid or base is added to the buffer it will be neutralized and not significantly change the pH of the buffer.
Uses of Buffers
Buffers keep things at a specific pH to allow them function correctly and be used efficiently Buffers are found in many places, including inside us. Our bodies create a buffer of carbonic acid and bicarbonate to create a pH between 7.35 and 7.45 to ensure that our blood plasma can function. Buffers are also found in more industrial settings, such as fermentation and setting dyes.
Buffers are not infinite though and can reach capacity. If enough acid or base is added to a buffer it cannot neutralize it all and it becomes ineffective.
A buffer is a chemical solution that is resistant to changes in its pH as its pH is very stable.
What makes up a Buffer and how does it work?
A buffer is made by adding large volumes of a weak acid or base to its conjugate acid or base as they can be in the same solution with out neutralizing each other. This ensures that if any acid or base is added to the buffer it will be neutralized and not significantly change the pH of the buffer.
Uses of Buffers
Buffers keep things at a specific pH to allow them function correctly and be used efficiently Buffers are found in many places, including inside us. Our bodies create a buffer of carbonic acid and bicarbonate to create a pH between 7.35 and 7.45 to ensure that our blood plasma can function. Buffers are also found in more industrial settings, such as fermentation and setting dyes.
Buffers are not infinite though and can reach capacity. If enough acid or base is added to a buffer it cannot neutralize it all and it becomes ineffective.