Strength vs. Concentration
Strength of acids/bases
So what exactly determines the strength of an acid? That would be its ability to lose its H+. In sciency words:
A strong acid is an acid that dissociates completely in water to form hydrogen ions. A weak acid is one dissociates only partially to form hydrogen ions in water.
Dissociation just means separation. So the more easily it can lose those ions the stronger it is. If the acid does not dissociate completely it will keep some sort of bond with the ions.
Now what about in a base? It's pretty much the same thing but you have OH- ions dissociating. The more complete that dissociation is the stronger the base it. Definition time:
A strong base is a base that dissociates completely in water to form hydroxide ions. A weak base is one dissociates only partially to form hydroxide ions in water.
Concentration
Concentration is defined as the number of particles per unit volume with the unit most often being a liter. It is very possible to have a concentrated amount of a weak acid or a diluted amount of a strong acid. While this changes how strong/ effective an acid seems it is important to remember that changing the concentration does not change the strength!
So what exactly determines the strength of an acid? That would be its ability to lose its H+. In sciency words:
A strong acid is an acid that dissociates completely in water to form hydrogen ions. A weak acid is one dissociates only partially to form hydrogen ions in water.
Dissociation just means separation. So the more easily it can lose those ions the stronger it is. If the acid does not dissociate completely it will keep some sort of bond with the ions.
Now what about in a base? It's pretty much the same thing but you have OH- ions dissociating. The more complete that dissociation is the stronger the base it. Definition time:
A strong base is a base that dissociates completely in water to form hydroxide ions. A weak base is one dissociates only partially to form hydroxide ions in water.
Concentration
Concentration is defined as the number of particles per unit volume with the unit most often being a liter. It is very possible to have a concentrated amount of a weak acid or a diluted amount of a strong acid. While this changes how strong/ effective an acid seems it is important to remember that changing the concentration does not change the strength!

Hopefully this is your reaction right about now.
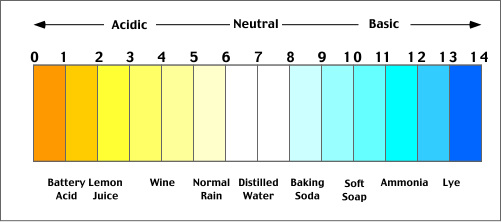
For clarity and future reference I've listed some example acids and bases broken down by their strength
|
Strong Acids
|
Weak Acids
|
Weak Bases
|
Strong Bases
|